NEUROMODULATION
Neuromodulation is the concept of stimulating and reorganizing nerve tissues that cause neurological diseases, have lost function, or cause various clinical symptoms due to dysfunction, through chemical or electrical applications. Providing stimulation in nerve tissues that have lost function can partially restore the missing function of this nerve tissue. With this method, abnormal stimuli and/or pain caused by nerve tissue can be suppressed with neuromodulation and the patient’s complaints can be reduced.
How does neuromodulation show its effect?
In nerve tissues that have suffered functional loss, a number of neurochemical substances are released and various abnormal electrical activities occur through these neurochemical substances. In a healthy nerve tissue, the nerve continues its physiological function with the type and help of these neurochemicals. Due to the damage to the nerve tissue and the functional loss that occurs as a result, physiological electrical activity may not be provided. In this respect, the electrical and chemical stimulation given to the patient from outside with neuromodulation provides the reorganization of these mechanisms.
For this stimulation, batteries that can generate electricity from outside or pumps that inject drugs help. These batteries used for neuromodulation are applied by placing electrodes into the tissue in a way that fits the nerve tissues in the brain, spinal cord or body areas close to these regions. These electrodes produce electrical impulses with an appropriate intensity to stimulate the targeted tissues. Problems related to many different neurological diseases such as Parkinson’s disease, essential tremor (tremor disease) and migraine can be solved with these methods.
What are the diseases in which neuromodulation is used?
Neuromodulation can be used in the treatment of any neurological disease that causes deterioration in the functions of nerve cells. Since neuromodulation equipment must be placed inside the body with interventional methods in patients and it is an expensive method, its application is usually brought to the agenda in clinical cases that are resistant to drug treatment, not suitable for surgical treatment and accompanied by severe clinical symptoms.
The most common disorders in which neuromodulation is used in clinical applications are epilepsy, migraine, chronic nerve-related pain, Parkinson’s disease, urinary incontinence and hearing loss. Neuromodulation treatment methods that are successfully applied today can be listed as follows:
- Spinal cord stimulation (spinal cord pacemaker)
- Posterior root ganglion stimulation
- Sacral nerve root stimulation (bladder pacemaker)
- Chronic pain neuromodulation
- Peripheral nerve stimulation
- Deep brain stimulation (brain pacemaker)
- Intrathecal drug infusion (baclofen pump)
- Vagal nerve stimulation (epilepsy pacemaker)
- Transcutaneous electrical nerve stimulation
- Transcranial magnetic stimulation
- Cochlear nerve stimulation
- Visual prosthesis
What are the concepts of neuromodulation and neurostimulation in migraine treatment?
Neuromodulation (NM) and neurostimulation (NS) have an important place among non-drug treatment options for migraine. Classic drug treatments are the first line in migraine treatment. The answer to the question of which migraine patients are subject to non-drug treatments is;
Patients whose attacks continue despite receiving appropriate drug treatment,
Patients who experience side effects due to the drugs used,
Patients who have interactions between drugs used for migraine treatment due to another disease,
Patients who have another medical condition that prevents them from taking appropriate drugs.
NM and NS have become important methods among non-drug treatment options for migraine.
How do neuromodulation and neurostimulation affect migraine?
The neurostimulation method can be briefly defined as the stimulation of loss of function or abnormal neurochemical response in the nerves associated with headache by means of an electrical current, magnetic current or direct current with the help of specially developed devices.
NS in migraine can be performed on the skin around the head, or with special devices placed invasively near the nerves that cause headaches.
Non-invasive methods applied on the skin around the head;
Supraorbital Transcutaneous Stimulation (STS)
Transcranial Magnetic Stimulation (TMS)
Transcranial Pulse Stimulation (TPS)
Transcranial Direct Current Stimulation (TDAU)
Vagal Nerve Stimulation (VSS)
Each of these methods may not be suitable for every migraine patient. Therefore, it should be determined which of these methods will be used selectively for patients whose appropriate indications have been determined by a neurologist.
Supraorbital Transcutaneous Stimulation (STS)
Cefaly
It is applied to the supraorbital nerve, which is also used for nerve blockade. It originates anatomically from the eye socket and is known to be associated with headaches. The device is attached to the head like a band or crown and provides electrical stimulation to the supraorbital nerve with certain frequencies.
The application practice is in the form of 20-minute stimulations per day for 3 months.
The first STS device approved by the American Food and Drug Administration (FDA) for chronic migraine was offered to patients under the name Cefaly᷿®. This device for chronic migraine was approved for prescription only, but later it was approved for over-the-counter sales.
This wearable migraine device has been updated as Cefaly Dual with the latest technological innovations and was approved by the FDA in 2020 for the acute (attack period) and preventive treatment of migraine headaches in adults aged 18 and over.
The device (Cefaly Dual) has 2 settings; ACUTE and PREVENT
In the ACUTE setting, the device is used for 60 minutes of pain relief at the beginning or during a migraine attack.
The PREVENT setting reduces the frequency and intensity of migraine symptoms in 20-minute sessions per day. The device automatically turns off at the end of each session.

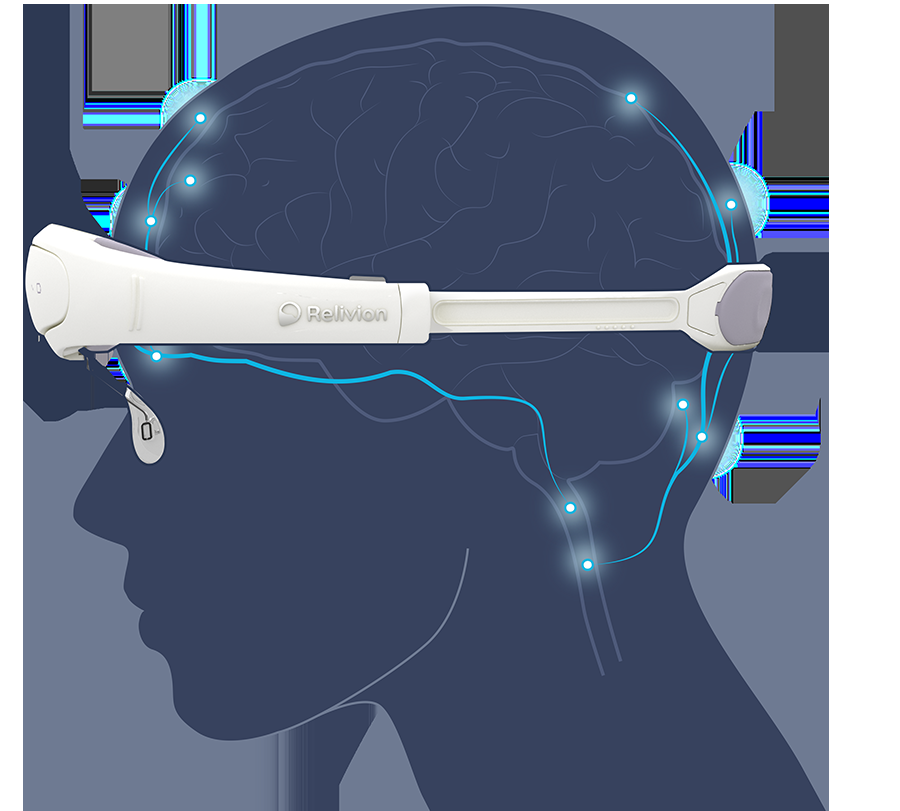
Relivion MG
Another e-TNS device is Relivion. Unlike Cefaly, this latest noninvasive device delivers electrical stimulation to both the trigeminal nerve branches and the occipital nerve branches (stimulating a total of 6 nerve branches). It has two sensors placed on either side of the nose. It was approved by the FDA in 2021 for the treatment of acute migraine in adults. It is in the device group that requires a doctor’s prescription.
Transcranial Magnetic Stimulation (TMS)
As its name suggests, it is a device that shows its effect by giving magnetic stimulation to certain brain regions. This device, which is more known among migraine patients, is thought to affect the release of certain neurochemicals by giving magnetic stimulation from the back of the head, known as the occipital region, with the help of an apparatus that we can call a stimulator, by affecting the electrical fields in the cerebral cortex. It is thought to achieve this effect by affecting the polarization in the nerve tissue.
The TMS device was approved by the FDA in 2013 for migraine patients aged 18 and over, only for migraine attacks with aura.
Having metal in the head, neck or upper body, having an active implanted medical device (pacemaker, deep brain stimulator etc.), having a diagnosis of epilepsy, having a history of seizures in the family or in oneself are contraindications for TMS.
As a rule, it should not be applied more than once in 24 hours.
Single-pulse TMS (sTMS)
A potential preventive and acute treatment for migraines, sTMS can be applied at home by placing and activating a device behind your head. The device delivers a short magnetic pulse (the length is preset by your doctor) that targets the layers of the scalp, skull, meninges (membranes surrounding the brain and spinal cord), cerebrospinal fluid, and superficial layers of the cortex, where it modulates the electrical environment of neurons involved in migraine attacks.
In a study published in the journal Cephalalgia in May 2018, sTMS reduced the number of days people with migraines experienced symptoms. Users also needed less rescue medication.
The eNeura sTMS mini is an FDA-approved sTMS device, previously sold under the brand name Spring TMS. Battery-powered and handheld, it can be used for both prevention and treatment of migraine attacks.
The user places and holds the sTMS mini firmly against the back of the head to locate the base of the skull. With the push of a button, the device’s specially shaped electrical coils send a magnetic pulse designed to treat migraine attacks and prevent them by interrupting the abnormal electrical activity in the brain that is associated with them.
You need a doctor’s prescription to use the sTMS mini.

Transcranial Pulse Stimulation
The device on the market under the name Neurolith® has a mechanotransduction mechanism. Although it is mainly used for Alzheimer’s disease (by increasing growth factors, VEGF, cerebral blood flow, new vessel formation, nerve regeneration and nitric oxide release), studies are also ongoing for the treatment of migraine.
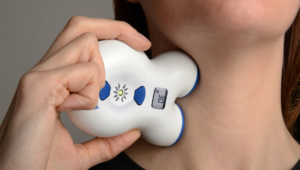
Vagal Nerve Stimulation (VSS)
Noninvasive VSS is thought to be effective in the treatment and prevention of migraine and cluster headache with the help of special devices such as the gammaCore® (electroCore) device. After being approved for the acute attack period of migraine in 2015, it was also licensed for the treatment of cluster headache in 2019.
The device has also been licensed for the acute and preventive treatment of migraine and cluster headaches for ages 12-17 as of February 2021. The device, which is the size of a computer mouse, applies a lubricating gel to the two stimulation points and then electrically stimulates the patient under the jawline, from any neck area for approximately 2 minutes. The intensity of the stimulation can be adjusted by the patient themselves. Regular use twice a day is recommended as a preventive treatment (for 3 months). It is the only NS device that has been approved for the treatment of hemicrania continua and paroxysmal hemicrania. A doctor’s prescription is required.
REN/Remote Electrical Neuromodulation
Remote electrical neuromodulation (REN) may be particularly useful for people with migraines who are concerned that using devices that apply electrical stimulation to the head may worsen the pain associated with attacks. Uniquely, this approach is designed to stimulate peripheral nerves in the upper arm. It is thought to reduce migraine pain by modulating a deep part of the nervous system involved in pain control. Specifically, REN activates pain control centers in the brainstem (the connection between the brain and spinal cord), which in turn blocks the pain signal that occurs in migraines.
Nerivio
Nerivio is a wireless remote neuromodulation armband that is FDA-approved for the acute treatment of migraine with or without aura in people ages 12 and older. The device is controlled by an app designed to provide personalized treatments.
The app includes a migraine diary that allows you to track treatment sessions and migraine attacks. You can share the diary with your doctor to help guide your migraine management.
Nerivio is available only with a doctor’s prescription.
Each Nerivio device provides twelve 45-minute treatments. After 12 treatments, the battery no longer works and the device must be recycled.

Implantable Occipital Nerve Stimulation
Implantable occipital nerve stimulation involves surgically implanting a device that sends electrical pulses to the occipital nerve, which is located at the back of the head, just above the neck. It is primarily used for chronic migraine patients who have failed other treatments.
A study published in the December 2020 issue of the journal Pain and Therapy found that the approach was effective for about 50 percent of people with chronic migraines who had an ONS device implanted.
ONS, in particular, differs from other nerve stimulation systems in that it involves surgery, but the procedure is reversible.
An implantable device under development by Salvia Bioelectronics uses mild electrical impulses to affect nerve activity, or the pattern of electrical impulses transmitted through the nerve fibers that control how your body works. The Dutch company is working on thin implantable bioelectronic foils that conform to the shape of your head to provide neurostimulation.
Specifically, the company’s implantable neurostimulation system (similar to a pacemaker) will be designed to use bioelectronic foil technology to disrupt brain processes that cause migraine attacks in people who have failed drug therapy. In 2020, the FDA granted Salvia a breakthrough device designation for its implantable technology.

- GammaCore
- NERİVİO
- CEFALY DUAL
- eNeura S-TMS mini
- RELİVİON

Cevapla
Want to join the discussion?Feel free to contribute!